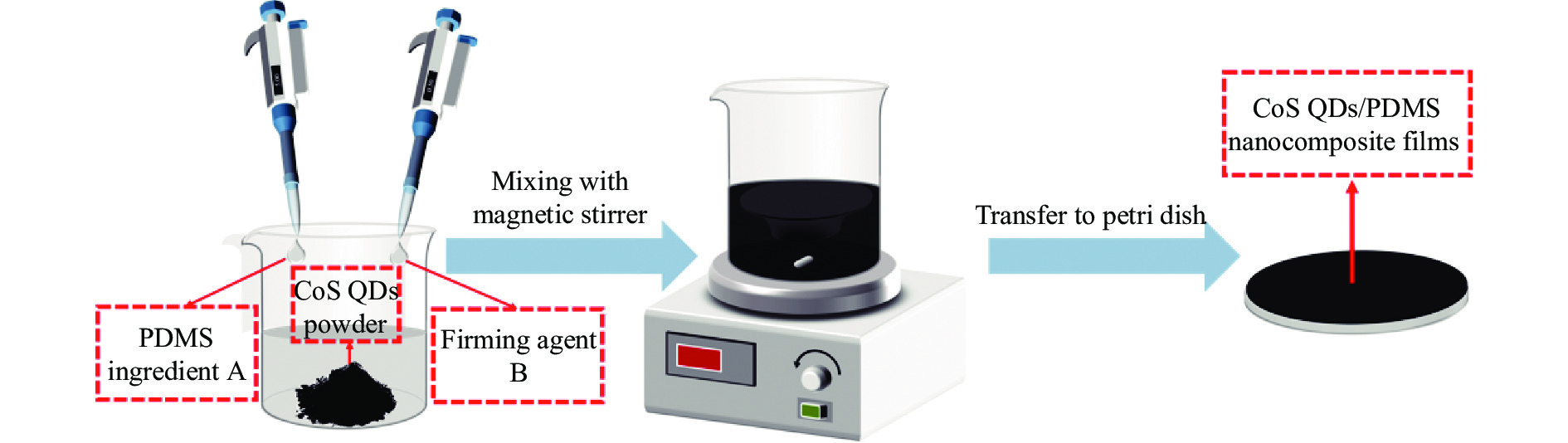
-
近年来,TMCs半导体材料由于其突出的物理、化学特性而受到越来越多的关注[1]。作为TMCs家族中的重要成员,CoS具有稳定的结构,且毒性比传统半导体材料低[2]。CoS晶体属六方晶系,Co-S键长为2.32 Å (1 Å=0.1 nm),空间群为P63/mmc (194),有着良好的对称性。由于CoS QDs禁带宽度较窄因而具有优异的近红外吸收特性,引起了人们的研究兴趣,并将其应用于光催化、太阳电池、激光器等领域。Joshi等[3]采用微波辐射法合成了平均粒径为58.28 nm,带隙为1.69 eV的CoS纳米颗粒(CoS NPs),该颗粒在200~2 000 nm波长范围内具有较强的吸收特性;Rani等[4]采用水热法合成了粒径为22 nm的CoS NPs,通过紫外-可见(UV-Vis)和PL测试发现当激发波长为330 nm时,该CoS NPs在360~580 nm波长范围内有发光现象,且在近红外区域具有吸收特性;Pourahmad等[5]先通过水热法合成了具有近红外吸收特性的CoS NPs,其平均尺寸约为58 nm,然后再采用离子交换法制备了CoS/铝硅酸盐纳米光催化剂,研究结果表明:当CoS的质量分数为17%时,该光催化剂对亚甲基蓝的降解效率最高; Kumar等[6]采用水热法合成得到了带隙为1.27 eV,具有八角结构的花形CoS纳米材料,UV-Vis测试发现该材料在可见光及近红外区域均有较宽的吸收,基于该CoS制备的对电极可提高染料敏化太阳电池的电流密度;Kung等[7]通过两步法在掺氟氧化锡衬底上制备了一维CoS针状纳米棒阵列,研究表明该CoS纳米棒阵列具有优异的近红外吸收特性,可用于制作染料敏化太阳电池的对电极,在100 mW/cm2光照下,CoS针状纳米棒阵列电池的最大功率转换效率为7.67%;Hui等[8]利用液相剥离法制备得到厚度为2 nm的CoS 纳米片,在200~1900 nm波长范围内具有宽带吸收,并基于该纳米片制备了调制深度为6.4%的新型CoS 纳米片可饱和吸收体,成功应用于锁模光纤激光器。
为进一步提升CoS纳米材料的光学特性和加工性能,CoS纳米薄膜及CoS纳米复合物的研究得到了广泛关注。Ighodalo等[9]采用化学沉积法成功制备得到了具有不同晶粒尺寸的CoS纳米薄膜,经研究发现这些薄膜的直接带隙在1.9~2.2 eV之间,平均光导率为1.77×1017 S,随着沉积循环次数的增加,CoS纳米薄膜在近红外区域的吸收逐渐增强,但薄膜的结晶度却逐渐降低,在大约50次沉积循环后,薄膜呈现出非晶态。Li等[10]将所制备的CoS纳米片与聚乙二醇(PEG)混合得到了CoS-PEG纳米片,该复合材料具有强烈的近红外吸收特性和优异的光热稳定性; Ding[11]等通过原位还原硫化/碳化2-甲基咪唑钴盐(ZIF-67)制备了粒径小于5 nm的CoS QDs,然后与氮掺杂的碳纳米管(CNTs)结合形成了CoS QDs/CNTs复合材料,UV-Vis测试表明该复合材料在200~800 nm波长范围内有吸收,表现出优异的近红外特性,且在短时间内可实现对活性氧的100%降解。
在不同CoS纳米材料中,CoS QDs因禁带宽度较窄、量子尺寸效应显著,具有载流子浓度高、光吸收系数大等特性,应用前景广泛。为了减少量子点应用过程中的聚集,将其与有机聚合物复配能够有效提升量子点的稳定性及加工性能,利于获得高效稳定的发光器件[12]。聚二甲基硅氧烷(PDMS)是一种具有高透明性、高柔韧性及高安全性的有机聚合物,常用作复合材料的配体。液相超声剥离法是制备纳米材料的一类常见方法,其操作简便、成本低廉、安全可控。因此,文中采用液相超声剥离法制备了CoS QDs,再通过共混法将CoS QDs与 PDMS复配得到CoS QDs/PDMS纳米复合薄膜,并对CoS QDs和CoS QDs/PDMS纳米复合薄膜的红外光学特性进行了研究,以期拓展CoS纳米复合薄膜材料在红外光学领域的应用。
-
实验采用液相超声剥离法制备CoS QDs溶液,制备步骤如下:称取0.15 g CoS粉末(纯度≥99.5%)放至研钵中充分研磨2 h;将研磨后的CoS粉末与50 mL无水乙醇(纯度≥99.7%)分散剂混合均匀,并将其置于超声仪中以90 W的功率累计超声2 h;将超声后的溶液进行离心,转速500 r/min,时间5 min;取出上层清液,即得到CoS QDs溶液;将CoS QDs溶液进行干燥处理备用。
-
实验采用共混法制备CoS QDs/PDMS纳米复合薄膜,制备步骤如图1所示。取5 mL PDMS的基本组分 A 和0.5 ml固化剂 B 移至烧杯中,并加入0.05 g 经干燥处理的CoS QDs粉末,使用磁力搅拌器搅5 min,随后转移至培养皿中,以30 ℃加热干燥直至成膜。
-
使用透射电镜(TEM, Tecnai G2 TF30 S-Twin)、原子力显微镜(AFM, 日本精工SPA-400)以及能谱仪 (EDS, NOVA NANOSEM 450)对CoS QDs的尺寸、形貌、结构及元素组分进行表征;使用X射线光电子能谱 (XPS, PHI Versa 探针 II)、X射线衍射仪(XRD, EMPYREAN, X射线源:Cu Ka,λ=0.154178 nm)、傅里叶变换红外光谱仪 (FTIR, Nicolet iS10)和拉曼光谱仪(Raman Renishaw-InVia)对CoS QDs的物相组成及成键特性进行分析;使用紫外-可见分光光度计 (UV-Vis, Shimadzu UV-3600 Puls)和荧光光谱仪 (PL&PLE, Hitachi, F-4500)对CoS QDs和CoS QDs/PDMS纳米复合薄膜的光学性质进行测试。
-
图2(a)为CoS QDs的TEM图和粒径分布直方图,可以看出CoS QDs具有良好的分散性,粒径符合正态分布,高斯拟合得到CoS QDs平均粒径约为5 nm,半峰宽(FWHM)为2 nm;图2(b)为CoS QDs的高分辨率TEM图,对其中的量子点使用Line Profile进行分析,得出晶格间距d=0.168 nm、d=0.193 nm、d=0.254 nm和d=0.257 nm,并分别对应于(1 1 0)、(1 0 2)、(1 0 1)和(0 0 2)晶面;图2(c)为在CoS QDs的高分辨率TEM图中选取其中一个量子点进行快速傅里叶变换分析(FFT),观察到CoS QDs呈现六方晶型,表明CoS QDs与CoS体材料的晶体结构相同。图2(d)为CoS QDs的AFM测试图,可以观察到CoS QDs尺寸均一、分布均匀,从中随机选取3个量子点,并标记为1、2、3进行粒径分析得到高度分别为4.9 nm、5.0 nm和5.1 nm,如图2(e)所示,该结果与TEM的粒径分析相吻合,表明CoS QDs呈球形。图2(f)为CoS QDs的EDS元素分析图,EDS能谱用于定性分析CoS QDs中元素的相对含量,在去除测试时包含的Cu、C等元素的干扰后得到Co元素(50.5%)与S元素(49.5%)的原子比例接近1∶1,符合CoS的1个Co原子与1个S原子相结合的结构模型。
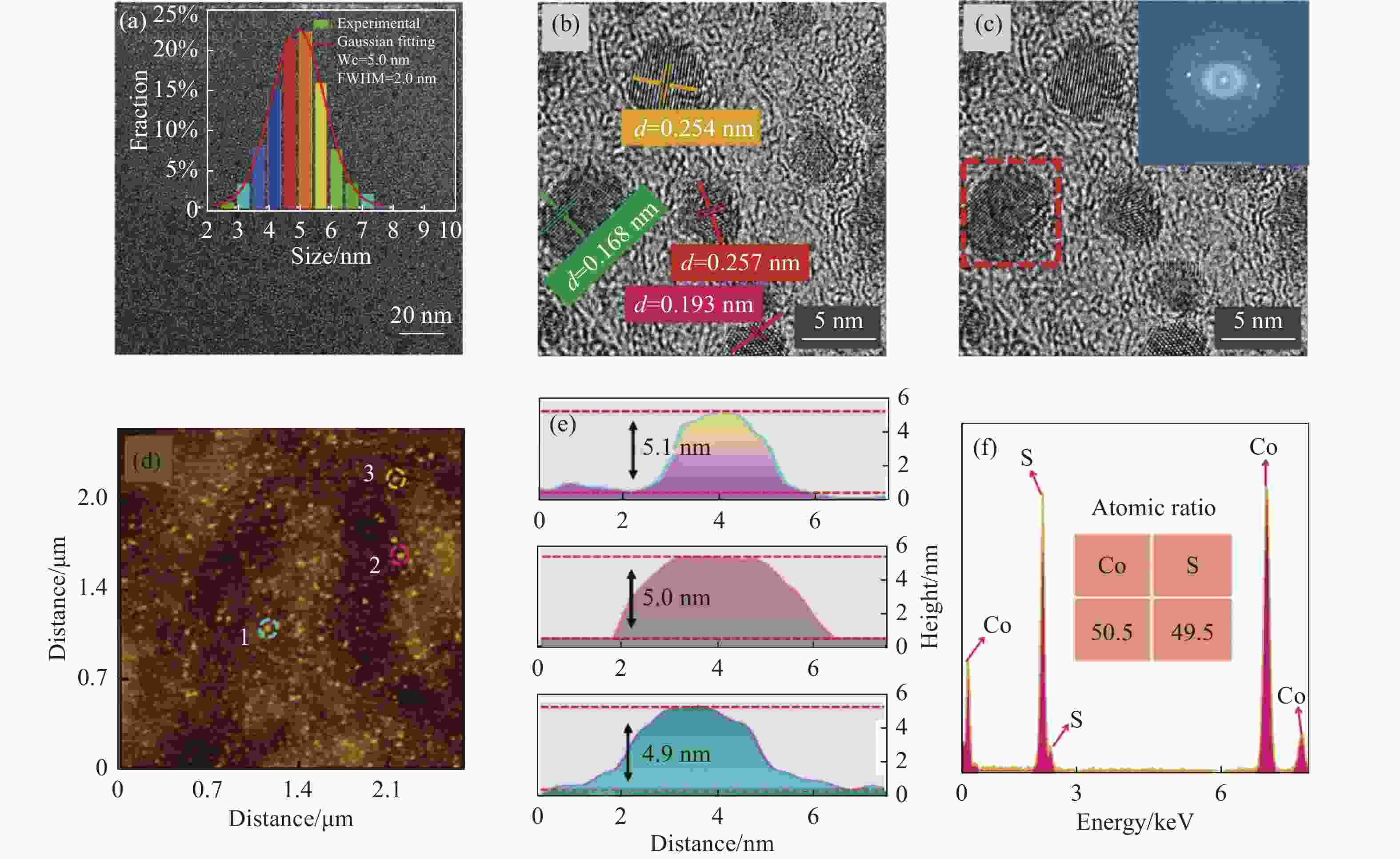
图 2 CoS QDs形貌、组分分析。(a) TEM图(插图为粒径分布图);(b) HR-TEM晶格条纹图;(c) HR-TEM图(插图为FFT图);(d) AFM图;(e) 在图(d)中标注的1、2、3号位置处的粒径高度分析图;(f) CoS QDs的EDS能谱图
Figure 2. The morphology and component analysis of CoS QDs. (a) TEM image (inset shows the particle size distribution); (b) HR-TEM image; (c) HR-TEM image (inset shows the FFT pattern); (d) AFM image; (e) Height analysis of the particle size at positions 1, 2 and 3 marked in Fig.(d); (f) EDS energy spectrum of CoS QDs
-
图3(a)为CoS QDs的XPS全谱图,从图中可以清晰地看到Co 2p、S 2p、C 1s和O 1s的XPS峰,其中Co 2p和S 2p是CoS的特征峰,C 1s可能是无水乙醇分散剂的残余,O 1s可能是样品CoS QDs表面被氧化或是无水乙醇分散剂的残余;为了深入分析CoS QDs中Co 2p和S 2p的存在形式,进行XPS分峰拟合。Co 2p的XPS谱如图3(b)所示,共有7种类型的峰,分别为Co 2p3/2(772.6 eV)、Co 2p3/2(778.8 eV)、Co 2p3/2(781.2 eV)、Co 2p3/2(783.6 eV)、Co 2p1/2(794.1 eV)、Co 2p1/2(797.1 eV)和Co 2p1/2(802.9 eV),其中前面6个峰都属于特征峰,772.6 eV、778.8 eV、794.1 eV对应Co3+[13], 781.2 eV、783.6 eV和797.1 eV对应Co2+[14],而802.9 eV对应的峰为卫星峰[15-16]。S 2p的XPS谱如图3(c)所示,共有3种类型的峰,分别为S 2p3/2(161.8 eV)、S 2p1/2(163.4 eV)和S 2p3/2(169.2 eV),其中161.8 eV和163.4 eV处出现的峰表明S是以S2−存在[17-19],而169.2 eV处出现的峰表明有S-O键存在[20],这可能是CoS表面的硫被氧化所导致的[13]。
FTIR测试结果如图3(d)所示,在528 cm−1和627 cm−1处分别观察到Co-S 键和CoS表面钴原子伸缩振动峰[21],其他振动为无水乙醇或无水乙醇氧化后所含的C、H和O所结合的化学键。CoS QDs的XRD测试结果如图3(e)所示,经过与标准PDF卡片(PDF#75-0605)对比可知,图中2θ=30.542° (d=0.29246 nm)、2θ=35.262°(d=0.25431 nm)、2θ=46.977° (d=0.19326 nm)、2θ=54.283° (d=0.16885 nm) 和 2θ=74.569° (d=0.12716 nm)处的衍射峰分别对应于CoS样品的(1 0 0)、(1 0 1)、(1 0 2)、(1 1 0)和(2 0 2)晶面,图中没有出现其他衍射峰,表明样品纯度高;CoS QDs的Raman测试结果如图3(f)所示,可以看到有4个清晰的Raman特征峰:189 cm−1、472 cm−1、512 cm−1和678 cm−1,分别对应硫化钴体材料的Ag、Eg、F2g和A1g振动模式[14,22-24],表明CoS是切实存在的。
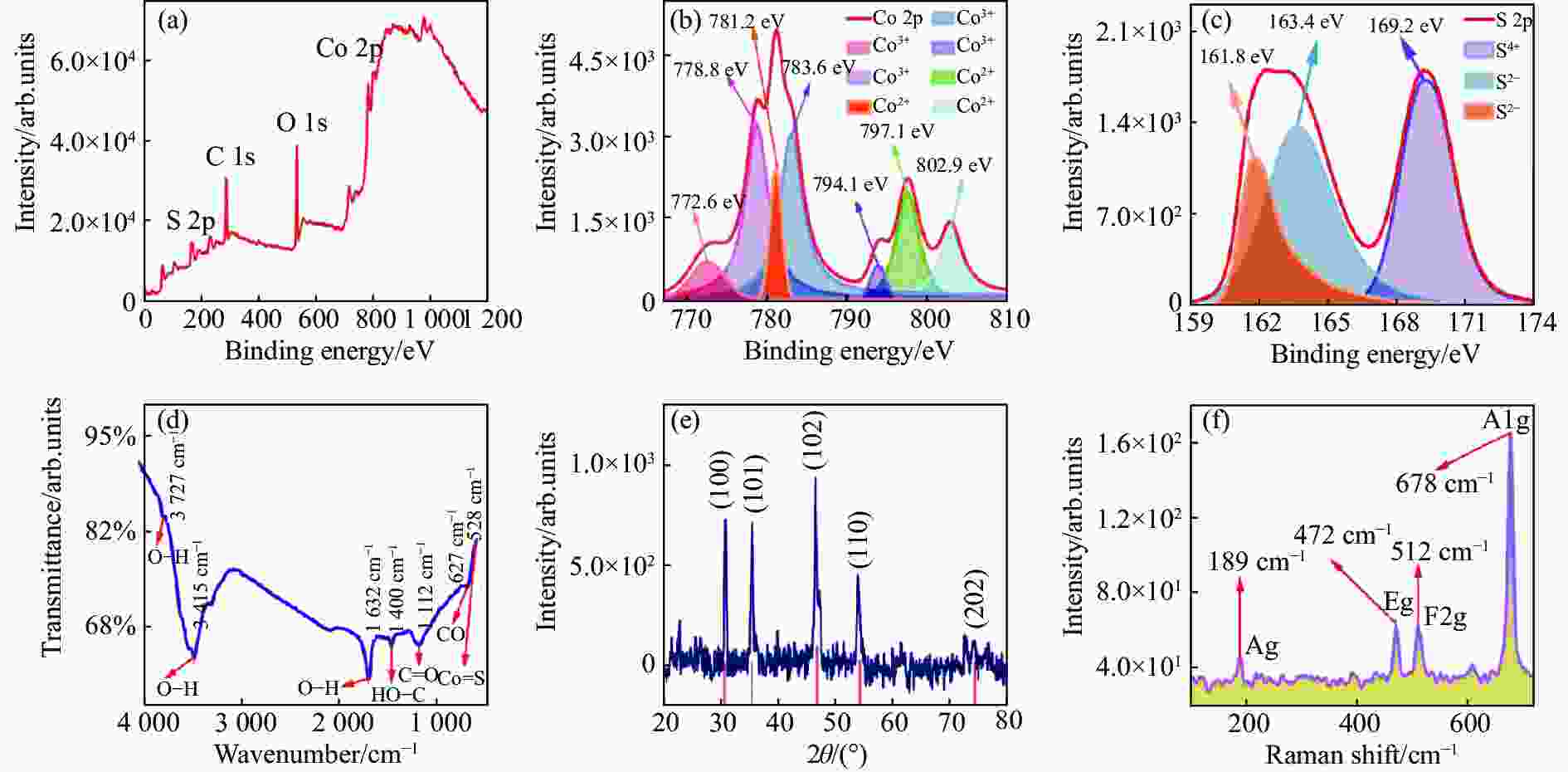
图 3 CoS QDs组分、化学键、物相、振动光谱分析。(a) XPS全谱图;(b) Co 2p XPS谱;(c) S 2p XPS谱;(d) FTIR谱;(e) XRD衍射图;(f) Raman谱
Figure 3. The component, chemical bond, phase, and vibrational spectrum analysis of CoS QDs. (a) XPS full spectrum; (b) Co 2p XPS spectrum; (c) S 2p XPS spectrum; (d) FTIR spectrum; (e) XRD diffraction pattern; (f) Raman spectrum
-
CoS QDs溶液的UV-Vis吸收光谱测试结果如图4(a)所示,该图表明CoS QDs从紫外到红外波段(200~2 000 nm)都有较强的吸收,且样品在相同浓度下吸收强度基本保持不变,随着样品稀释倍数的增加吸收强度逐渐降低。图4(a)中的插图则表明CoS QDs溶液具有荧光效应:样品在自然光照射下呈现无色,在波长为254 nm和356 nm的紫外光照射下,分别呈现出淡紫色和蓝色。为了检测纯CoS QDs薄膜的吸收特性,以石英为衬底将 CoS QDs 溶液制备成膜进行测试(图4(b)),由此可以看出,CoS QDs薄膜在紫外到红外波段(200~2 000 nm)同样具有吸收,但整个区间的吸收强度随波长增加呈下降趋势。CoS QDs/PDMS纳米复合薄膜的UV-Vis吸收光谱测试结果如图4(c)所示,插图是自然光照下的PDMS 薄膜(左)和 CoS QDs/PDMS纳米复合薄膜(右)的照片。图中下方的橙色曲线是PDMS薄膜的吸收,可以看到PDMS薄膜本身在200-2200 nm波长范围内基本没有吸收;红色曲线为CoS QDs/PDMS纳米复合薄膜的UV-Vis吸收,可以看到该纳米复合薄膜从紫外到红外波段(200~2200 nm)都具有强烈吸收, 且整个区间的吸收强度随波长的增加而缓慢增加,表明CoS QDs与PDMS复合成膜以后,红外吸收特性得到有效增强。这是因为复合薄膜中的CoS QDs浓度较高,从而表现出较强的红外吸收特性;图中蓝色曲线表示放置六个月的CoS QDs/PDMS纳米复合薄膜吸收曲线,可以看出经过长时间放置的薄膜其UV-Vis吸收强度几乎没有发生变化,光学性质十分稳定。
图4(d)是CoS QDs的PL光谱图,从图中可以看到,当激发光波长从360 nm增加到500 nm (步长为20 nm)时,CoS QDs的PL峰逐渐红移。经归一化处理的PL光谱如图4(e)所示,该图更直观地反映了这一红移现象,其PL峰值所对应的波长变化为:506.4 nm→517.6 nm→522 nm→523.6 nm→528.4 nm→533.4 nm→539.2 nm→545.6 nm,通过CoS QDs的PL峰红移可以看出激发波长会直接影响CoS QDs的光致发光。图4(f)是CoS QDs在近红外波段的PL光谱图,当激发波长以50 nm的步长从550 nm增加至750 nm时,从图4(f) 内嵌的放大图可以清晰地看到CoS QDs在近红外波段仍存在发光现象。图4(g)是CoS QDs的光致发光激发(PLE)光谱图,从图中可以看到,当发射光波长在420-550 nm范围内依次增加时,CoS QDs的PLE峰也出现类似PL峰的红移现象。归一化处理的PLE光谱图如图4(h)所示,其PLE峰值所对应的波长变化为:368.4 nm→373.6 nm→380 nm→393.6 nm→396.2 nm→406.2 nm;图4(i)是CoS QDs在近红外波段的PLE光谱图,当发射光波长从600 nm增加至750 nm时(步长为50 nm),从图4(i)中的插图可以看到CoS QDs在近红外波段仍存在响应。通过上述PL和PLE光谱分析可知CoS QDs溶液的发光具有波长依赖性,并且存在明显的Stokes位移效应。

图 4 CoS QDs及其PDMS纳米复合薄膜红外特性分析。(a) CoS QDs溶液的UV-Vis吸收光谱图(插图:自然光和紫外光照射下的CoS QDs溶液照片);(b) CoS QDs薄膜的UV-Vis吸收光谱图;(c) PDMS与CoS QDs/PDMS纳米复合薄膜的UV-Vis吸收光谱图;(d) CoS QDs溶液的PL谱图;(e) 归一化的CoS QDs的PL谱图;(f) 近红外CoS QDs的PL谱图;(g) CoS QDs的PLE谱图;(h) 归一化的CoS QDs的PLE谱图;(i) 近红外CoS QDs的PLE谱图;(j) PDMS的PL谱图;(k) CoS QDs/PDMS纳米复合薄膜的PL谱图;(l) 近红外区域CoS QDs/PDMS纳米复合薄膜的PL谱图
Figure 4. Infrared characterization of CoS QDs and their PDMS nanocomposite films. (a) UV-Vis absorption spectra of CoS QDs solutions (inset: photos of CoS QDs solutions under natural and UV light); (b) UV-Vis absorption spectra of CoS QDs films; (c) UV-Vis absorption spectra of CoS QDs of PDMS and CoS QDs/PDMS nanocomposite films; (d) PL spectra of CoS QDs solution; (e) PL spectra of normalized CoS QDs; (f) PL spectra of NIR CoS QDs; (g) PLE spectra of CoS QDs; (h) PLE spectra of CoS QDs; (i) PLE spectra of NIR CoS QDs; (j) PL spectra of PDMS plots; (k) PL spectra of CoS QDs/PDMS nanocomposite films; (l) PL spectra of near-infrared CoS QDs/PDMS nanocomposite films
图4(j)和(k)分别是PDMS薄膜和CoS QDs/PDMS纳米复合薄膜的PL光谱图,从图4(j)中可以看出,当激发光波长从320 nm逐渐增加到400 nm (步长20 nm)时,PDMS薄膜仅现了微弱的PL峰,并且随着激发波长的增加PDMS薄膜的发光强度逐渐降低;而图4(k)中的CoS QDs/PDMS纳米复合薄膜则具有明显的发光峰,说明在相同的波长激发下CoS QDs/PDMS纳米复合薄膜获得的能量更多,且出现的PL峰呈现出明显的红移现象。但与CoS QDs溶液相比,纳米复合薄膜的发光强度有所减弱,这可能是因为复合薄膜中的CoS QD浓度较高发生了浓度淬灭,从而导致纳米复合薄膜发光强度降低。图4(l)是CoS QDs/PDMS纳米复合薄膜在近红外波段的 PL 图,当激发光波长以50 nm的步长从600 nm增加至750 nm时,从图4(l)的插图可以看到CoS QDs/PDMS纳米复合薄膜在近红外波段(620-900 nm)仍具有发光现象。
-
文中通过液相超声剥离法成功制备出了具有分散性良好、颗粒大小均匀、平均粒径约为5 nm的球形CoS QDs,并采用共混法将CoS QDs与PDMS混合制备出了CoS QDs/PDMS纳米复合薄膜;通过UV-Vis测试发现CoS QDs溶液从紫外到红外波段(200~2 000 nm)都有吸收,且吸收强度随样品浓度的降低而降低;CoS QDs/PDMS纳米复合薄膜从紫外到红外波段(200~2200 nm)都具有吸收,与CoS QDs薄膜相比,CoS QDs/PDMS纳米复合薄膜的红外吸收特性得到有效增强,并且薄膜样品放置六个月后吸收强度几乎没有发生变化;通过PL测试发现CoS QDs和CoS QDs/PDMS纳米复合薄膜在红外波段均具有光致发光现象,PL峰有明显红移现象,存在 Stokes 位移效应,两者的发光都具有波长依赖性。综上,基于CoS QDs制备的CoS QDs/PDMS纳米复合薄膜具有优异的光学特性,尤其是红外波段的吸收和发光特性尤为瞩目,且光学性质稳定,表明该复合材料在红外探测器、纳米光子器件、柔性显示器、红外激光器等领域具有重要的潜在应用价值,值得进一步探索和研究。
Study on preparation and infrared properties of CoS QDs/PDMS nanocomposite films
-
摘要: 过渡金属硫属化合物(TMCs)由于具有优异的光学、电学及光电等特性,被广泛应用于光催化、太阳电池、激光器等领域。作为一类典型的TMCs材料,硫化钴量子点(CoS QDs)因禁带宽度较窄而具有优异的近红外吸收特性,有望用于红外技术领域。文中采用液相超声剥离法制备了CoS QDs,再用共混法制备得到CoS QDs/PDMS纳米复合薄膜,并对它们的光学性质进行了研究,结果表明:CoS QDs的平均尺寸约为5 nm,大小均匀,呈球形;CoS QDs 与CoS QDs/PDMS纳米复合薄膜在红外波段均存在明显的吸收和发光特性,且复合薄膜的红外吸收特性优于CoS QDs薄膜;随着激发光波长的增加,纳米复合薄膜的光致发光(PL)峰出现了红移,表现出明显的Stokes位移效应和激发波长依赖性。CoS QDs/PDMS纳米复合薄膜优异的红外吸收和发光特性,表明其在红外探测、荧光成像、纳米光子器件等研究领域中具有重要的潜在应用价值,有望成为一种新型红外探测材料。Abstract:
Objective TMCs have been widely used in photocatalysis, solar cells, lasers and other fields because of their excellent optical, electrical and photoelectric properties. As a typical TMCs material, CoS QDs have excellent near-infrared absorption properties due to their narrow band gap and are expected to be used in infrared technology. CoS QDs are expected to be an important material for infrared detector preparation. In order to improve the optical properties and processing properties of CoS QDs, CoS QDs were further prepared into nanocomposite films to expand their application range. At present, the research work on CoS mainly focuses on CoS NPs, and there are few reports on quantum dot composite films. Therefore, the CoS QDs prepared by liquid phase ultrasonic exfoliation method are blended with PDMS, and the infrared properties of CoS QDs /PDMS nanocomposite films are studied. In order to expand the application of CoS QDs in infrared optics. Methods CoS QDs solution was prepared by liquid phase ultrasonic exfoliation method. The preparation steps were as follows: 0.15 g CoS powder (purity ≥99.5%) was weighed and put into a mortar and fully ground for2 h; The ground CoS powder was evenly mixed with 50 mL of anhydrous ethanol (purity ≥99.7%) dispersant, and placed in the ultrasonic instrument at 90 W power for 2 h; The ultrasonic solution was centrifuged at a rotational speed of 500 r/min for 5 min. Taking out the supernatant, CoS QDs solution was obtained. The CoS QDs solution is dried for later use. CoS QDs/PDMS nanocomposite films were prepared by blending method. 5 mLof the basic component A of PDMS and 0.5 mL of the curing agent B were transferred to the beaker, and appropriate amount of the dried CoS QDs powder was added, stirred with a magnetic stirrer for 5 min, and then transferred to the petri dish and heated at 30 ℃ until film formation. The size, morphology, structure and elemental components of CoS QDs were characterized by transmission electron microscopy (TEM), atomic force microscopy (AFM) and energy dispersive spectrometry (EDS). The phase composition and bonding properties of CoS QDs were analyzed by X-ray photoelectron spectroscopy (XPS), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and Raman spectroscopy (Raman); the optical properties of CoS QDs and CoS QDs/PDMS composite films were tested by UV-Vis spectrophotometer and fluorescence spectrometer. Results and Discussions Both CoS QDs and CoS QDs/PDMS nanocomposite films have obvious absorption and luminescence characteristics in infrared band, and the infrared absorption characteristics of the composite films are better than that of CoS QDs films (Fig.4(a)-(c)). With the increase of excitation wavelength, the PL peak of CoS QDs/PDMS nanocomposite films shows a redshift, which shows obvious Stokes shift effect and excitation wavelength dependence (Fig.4(k)). Conclusions The spherical CoS QDs with good dispersion, uniform particle size and average particle size of about 5 nm were successfully prepared by liquid phase ultrasonic stripping method, and the CoS QDs/PDMS nanocomposite films were prepared by blending CoS QDs and PDMS. After UV-Vis test, it was found that CoS QDs solution and CoS QDs/PDMS nanocomposite films have absorption from ultraviolet to infrared band(200-2200 nm). Compared with CoS QDs films, the infrared absorption characteristics of CoS QDs/PDMS nanocomposite films are effectively enhanced. Moreover, the absorption strength of the film samples hardly changed after six months. The PL test shows that CoS QDs and CoS QDs/PDMS nanocomposite films have PL phenomenon in infrared band, PL peak has obvious redshift phenomenon, Stokes shift effect, and both have wavelength dependence. On the other hand, CoS QDs/PDMS nanocomposite films have excellent infrared optical properties, especially the absorption and luminescence characteristics in the infrared band, and the optical properties are very stable, indicating that the composite material has important potential application value in the fields of infrared detectors, nano-photonic devices, flexible displays, infrared lasers and so on. -
图 2 CoS QDs形貌、组分分析。(a) TEM图(插图为粒径分布图);(b) HR-TEM晶格条纹图;(c) HR-TEM图(插图为FFT图);(d) AFM图;(e) 在图(d)中标注的1、2、3号位置处的粒径高度分析图;(f) CoS QDs的EDS能谱图
Figure 2. The morphology and component analysis of CoS QDs. (a) TEM image (inset shows the particle size distribution); (b) HR-TEM image; (c) HR-TEM image (inset shows the FFT pattern); (d) AFM image; (e) Height analysis of the particle size at positions 1, 2 and 3 marked in Fig.(d); (f) EDS energy spectrum of CoS QDs
图 3 CoS QDs组分、化学键、物相、振动光谱分析。(a) XPS全谱图;(b) Co 2p XPS谱;(c) S 2p XPS谱;(d) FTIR谱;(e) XRD衍射图;(f) Raman谱
Figure 3. The component, chemical bond, phase, and vibrational spectrum analysis of CoS QDs. (a) XPS full spectrum; (b) Co 2p XPS spectrum; (c) S 2p XPS spectrum; (d) FTIR spectrum; (e) XRD diffraction pattern; (f) Raman spectrum
图 4 CoS QDs及其PDMS纳米复合薄膜红外特性分析。(a) CoS QDs溶液的UV-Vis吸收光谱图(插图:自然光和紫外光照射下的CoS QDs溶液照片);(b) CoS QDs薄膜的UV-Vis吸收光谱图;(c) PDMS与CoS QDs/PDMS纳米复合薄膜的UV-Vis吸收光谱图;(d) CoS QDs溶液的PL谱图;(e) 归一化的CoS QDs的PL谱图;(f) 近红外CoS QDs的PL谱图;(g) CoS QDs的PLE谱图;(h) 归一化的CoS QDs的PLE谱图;(i) 近红外CoS QDs的PLE谱图;(j) PDMS的PL谱图;(k) CoS QDs/PDMS纳米复合薄膜的PL谱图;(l) 近红外区域CoS QDs/PDMS纳米复合薄膜的PL谱图
Figure 4. Infrared characterization of CoS QDs and their PDMS nanocomposite films. (a) UV-Vis absorption spectra of CoS QDs solutions (inset: photos of CoS QDs solutions under natural and UV light); (b) UV-Vis absorption spectra of CoS QDs films; (c) UV-Vis absorption spectra of CoS QDs of PDMS and CoS QDs/PDMS nanocomposite films; (d) PL spectra of CoS QDs solution; (e) PL spectra of normalized CoS QDs; (f) PL spectra of NIR CoS QDs; (g) PLE spectra of CoS QDs; (h) PLE spectra of CoS QDs; (i) PLE spectra of NIR CoS QDs; (j) PL spectra of PDMS plots; (k) PL spectra of CoS QDs/PDMS nanocomposite films; (l) PL spectra of near-infrared CoS QDs/PDMS nanocomposite films
-
[1] Jing Y M, Liu B Z, Zhu X K, et al. Tunable electronic structure of two-dimensional transition metal chalcogenides for optoelectronic applications [J]. Nanophotonics, 2020, 9(7): 1675-1694. doi: 10.1515/nanoph-2019-0574 [2] Yin Z Z. Preparation of TiO2-based heterojunction and its photocatalytic performance of tetracycline hydrochloride [D]. Hefei: Anhui University, 2022. (in Chinese) [3] Joshi J H, Khunti D D, Joshi M J, et al. Penn model and wemple-didomenico single oscillator analysis of cobalt sulfide nanoparticles [J]. Functional Oxides and Nanomaterials, 2017, 1837: 040033. doi: 10.1063/1.4982117 [4] Rani B J, Pradeepa S S, Hasan Z M, et al. Supercapacitor and OER activity of transition metal (Mo, Co, Cu) sulphides [J]. Journal of Physics and Chemistry of Solids, 2020, 138: 109240. doi: 10.1016/j.jpcs.2019.109240 [5] Pourahmad A, Sohrabnezhad S, Radaee E, et al. Degradation of basic blue 9 dye by CoS/nanoAlMCM-41 catalyst under visible light irradiation [J]. Journal of Porous Materials, 2010, 17(3): 367-375. doi: 10.1007/s10934-009-9301-8 [6] Kumar K A, Pandurangan A, Arumugam S, et al. Effect of Bi-functional hierarchical flower-like CoS nanostructure on its interfacial charge transport kinetics, magnetic and electrochemical behaviors for supercapacitor and dssc applications [J]. Scientific Reports, 2019, 9: 1228. doi: 10.1038/s41598-018-37463-0 [7] Kung C W, Chen H W, Lin C Y, et al. CoS acicular nanorod arrays for the counter electrode of an efficient dye-sensitized solar cell [J]. ACS Nano, 2012, 6(8): 7016-7025. doi: 10.1021/nn302063s [8] Hui Z Q, Li N, Zhang X L, et al. CoS nanosheets for generation of vector soliton and bound solitons in nonlinear optical fiber system [J]. Optics and Laser Technology, 2023, 160: 109026. doi: 10.1016/j.optlastec.2022.109026 [9] Ighodalo K O, Ezealigo B N, Agbogu A, et al. The effect of deposition cycles on intrinsic and electrochemical properties of metallic cobalt sulfide by simple chemical route [J]. Materials Science in Semiconductor Processing, 2019, 101: 16-27. doi: 10.1016/j.mssp.2019.05.015 [10] Li Z L, Li Z, Chen L, et al. Polyethylene glycol-modified cobalt sulfide nanosheets for high-performance photothermal conversion and photoacoustic/magnetic resonance imaging [J]. Nano Research, 2018, 11(5): 2436-2449. doi: 10.1007/s12274-017-1865-z [11] Ding J, Zhu Y B, Ma Y L, et al. Spiderweb-inspired all-weather CoS quantum dots confined in n-doped carbon for boosted sulfate radical evolution [J]. Chemical Communications, 2022, 58(49): 6954-6957. doi: 10.1039/d2cc01523d [12] 陈星帆, 李斌, 李学铭, 等. 量子点-聚合物纳米复合材料的光电器件研究进展[J]. 红外与激光工程, 2022, 51(05): 416-430. doi: 10.3788/IRLA20210637 Chen Xingfan, Li Bin, Li Xueming, et al. Research advances in optoelectronic devices of quantum dot-polymer nanocomposites [J]. Infrared and Laser Engineering, 2022, 51(5): 20210637. (in Chinese) doi: 10.3788/IRLA20210637 [13] Chauhan M, Reddy K P, Deka S, et al. Copper cobalt sulfide nanosheets realizing a promising electrocatalytic oxygen evolution reaction [J]. ACS Catalysis, 2017, 7(9): 5871-5879. doi: 10.1021/acscatal.7b01831 [14] Shi J H, Li X C, He G H, et al. Electrodeposition of high-capacitance 3D CoS/graphene nanosheets on nickel foam for high-performance aqueous asymmetric supercapacitors [J]. Journal of Materials Chemistry A, 2015, 3(41): 20619-20626. doi: 10.1039/c5ta04464b [15] Pan Y, Liu Y Q, Liu C G, et al. Phase-and morphology-controlled synthesis of cobalt sulfide nanocrystals and comparison of their catalytic activities for hydrogen evolution [J]. Applied Surface Science, 2015, 357: 1133-1140. doi: 10.1016/j.apsusc.2015.09.125 [16] Liu H, You Z W, Yang S, et al. High-efficient adsorption and removal of elemental mercury from smelting flue gas by cobalt sulfide [J]. Environmental Science and Pollution Research International, 2019, 26(7): 6735-6744. doi: 10.1007/s11356-019-04159-5 [17] Yang L, Wu X L, Zhu X S, et al. Amorphous nickel/cobalt tungsten sulfide electrocatalysts for high-efficiency hydrogen evolution reaction [J]. Applied Surface Science, 2015, 341: 149-156. doi: 10.1016/j.apsusc.2015.03.018 [18] Tang J H, Ge Y C, Shen J F, et al. Facile synthesis of CuCo2S4 as a novel electrode material for ultrahigh supercapacitor performance [J]. Chemical Communications, 2016, 52(7): 1509-1512. doi: 10.1039/c5cc09402j [19] Wang X W, Batter B, Xie Y, et al. Highly crystalline, small sized, monodisperse α-NiS nanocrystal ink as an efficient counter electrode for dye-sensitized solar cells [J]. Journal of Materials Chemistry A, 2015, 3(31): 15905-15912. doi: 10.1039/c5ta02946e [20] Xu L, Lu Y. One-step synthesis of a cobalt sulfide/reduced graphene oxide composite used as an electrode material for supercapacitors [J]. RSC Advances, 2015, 5(83): 67518-67523. doi: 10.1039/c5ra11711a [21] Nan K K, Du H F, Su L, et al. Directly electrodeposited cobalt sulfide nanosheets as advanced catalyst for oxygen evolution reaction [J]. Chemistryselect, 2018, 3(25): 7081-7088. doi: 10.1002/slct.201801482 [22] Chen C Y, Shih Z Y, Yang Z S, et al. Carbon nanotubes/cobalt sulfide composites as potential high-rate and high-efficiency supercapacitors [J]. Journal of Power Sources, 2012, 215: 43-47. doi: 10.1016/j.jpowsour.2012.04.075 [23] Huang T Y, He M, Zhou Y M, et al. Solvothermal fabrication of CoS nanoparticles anchored on reduced graphene oxide for high-performance microwave absorption [J]. Synthetic Metals, 2017, 224: 46-55. doi: 10.1016/j.synthmet.2016.12.015 [24] Ren Y R, Wang H Y, Zhang T Y, et al. Designed preparation of CoS/Co/MoC nanoparticles incorporated in N and S dual-doped porous carbon nanofibers for high-performance Zn-air batteries [J]. Chinese Chemical Letters, 2021, 32(7): 2243-2248. doi: 10.1016/j.cclet.2020.12.050 -






 下载:
下载: